
Asquith Medicinal Chemistry Group
Leaders
Group Overview
The Asquith Medicinal Chemistry Group was established in late 2021 based at the School of Pharmacy, part of the University of Eastern Finland. The lab explores novel chemical strategies to improve the efficacy and safety of viral and cancer treatments. This includes development of highly selective inhibitors of kinases involved in cancer and host directed anti-virals. The focus is on the translation of fundamental and clinical research findings to real world application through a series of cutting-edge medicinal chemistry tools including prognostic solvation shell mapping, free energy perturbation (FEP) calculations and machine learning algorithms. These techniques coupling with a strong synthetic background enable us to use advanced medicinal chemistry as a central framework to translate research from early-stage discovery, through optimization, to real-world clinical benefit.
We have been integrally involved in projects towards the development of chemical probes for the under-studied kinases including GAK, AAK1, BIKE, STK10, SLK, CaMKK2, PKN3, ULK3, ADCK3/COQ8A, MAP4K4, TLK2 and PKMYT1. The aim of these efforts in part is to generate research tools to enable the elucidation of the biological functions of these kinases. This has resulted in four high-quality chemical probes for GAK ( UNC-CA93.0 / SGC-GAK-1 ) [1-2], EGFR ( UNC-CA359 ) [3-4], PKN3 ( UNC-CA94 ) [5], COQ8A ( TPP-UNC-CA157 ) [6]. These efforts have also laid the groundwork for the development of other chemical tools and probes for kinases including MAP4K4 ( UNC-CA409 ) [7], STK10 & SLK [8] and more recently CaMKK2 [9] leading to SGC-CaMKK2-1, and TLK2 ( UNC-CA2-103 ) [10].
Selected References
- Asquith, CRM et al. Identification and Optimization of 4-Anilinoquinolines as Inhibitors of Cyclin G Associated Kinase. ChemMedChem. 2018 , 13 , (1), 48-66. doi: 10.1002/cmdc.201700663.
- Asquith, CRM et al. SGC-GAK-1: A Chemical Probe for Cyclin G Associated Kinase (GAK). J. Med. Chem. 2019 , 62 , (5), 2830-2836. doi: 10.1021/acs.jmedchem.8b01213.
- Asquith, CRM et al. Design of a Cyclin G Associated Kinase (GAK)/Epidermal Growth Factor Receptor (EGFR) Inhibitor Set to Interrogate the Relationship of EGFR and GAK in Chordoma. J Med Chem. 2019 , 62 , (9), 4772-4778. doi: 10.1021/acs.jmedchem.9b00350.
- Bieberich, AA et al. Optimization of the 4-anilinoquin(az)oline scaffold as epidermal growth factor receptor (EGFR) inhibitors for chordoma utilizing a Toxicology profiling assay platform. Sci Rep. 2022 , 12 , (1), 12820. doi: 10.1038/s41598-022-15552-5.
- Asquith, CRM et al. Identification of 4-Anilinoquin(az)oline as a Cell-Active Protein Kinase Novel 3 (PKN3) Inhibitor Chemotype. ChemMedChem. 2022 , 17 , (12), e202200161. doi: 10.1002/cmdc.202200161.
- Murray, NH et al. Small-molecule inhibition of the archetypal UbiB protein COQ8. Nat Chem Biol. 2023 , 19 , (2), 230-238. doi: 10.1038/s41589-022-01168-3.
- Strang, BL et al. Identification of lead anti-human cytomegalovirus compounds targeting MAP4K4 via machine learning analysis of kinase inhibitor screening data. PLoS One. 2018 , 13 , (7), e0201321. doi: 10.1371/journal.pone.0201321.
- Asquith, CRM et al. Design and Analysis of the 4-Anilinoquin(az)oline Kinase Inhibition Profiles of GAK/SLK/STK10 Using Quantitative Structure-Activity Relationships. ChemMedChem. 2020 , 15 , (1), 26-49. doi: 10.1002/cmdc.201900521.
- Eduful, BJ et al. Hinge Binder Scaffold Hopping Identifies Potent Calcium/Calmodulin-Dependent Protein Kinase Kinase 2 (CAMKK2) Inhibitor Chemotypes. J Med Chem. 2021 , 64 , (15), 10849-10877. doi: 10.1021/acs.jmedchem.0c02274.
- Asquith, CRM et al. Discovery and Optimization of Narrow Spectrum Inhibitors of Tousled Like Kinase 2 (TLK2) Using Quantitative Structure Activity Relationships. Eur J Med Chem. 2024 , 271 , 116357. doi: 10.1016/j.ejmech.2024.116357.
News
-
 Kinase Target Watch: Article series highlights therapeutic opportunities within the understudied kinome
Kinase Target Watch: Article series highlights therapeutic opportunities within the understudied kinomeKinase Target Watch: Article series highlights therapeutic opportunities within the understudied kinome
This story highlights our work to shine a spotlight on understudied kinases in concise perspectives that have served to stimulate interest, guide… -
 The Jane and Aatos Erkko Foundation supports computer-assisted drug development for challenging drug targets
The Jane and Aatos Erkko Foundation supports computer-assisted drug development for challenging drug targetsThe Jane and Aatos Erkko Foundation supports computer-assisted drug development for challenging drug targets
This story highlights our collaborative funding towards new small molecules against small GTPase proteins associated with cancer and developmental… -
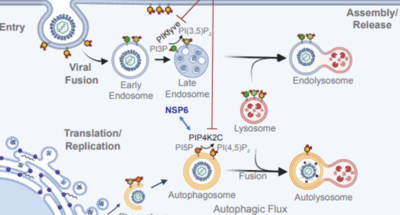 Emerging viral threats combatted by a potent new dual lipid kinase inhibitor
Emerging viral threats combatted by a potent new dual lipid kinase inhibitorEmerging viral threats combatted by a potent new dual lipid kinase inhibitor
This story highlights our collaborative study leading to a new anti-viral therapeutic strategy to simultaneously target lipid kinases PIKfyve and… -
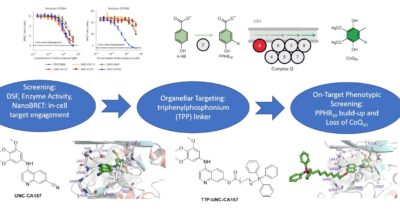 The first potent COQ8 inhibitor targets ubiquinone biosynthesis
The first potent COQ8 inhibitor targets ubiquinone biosynthesisThe first potent COQ8 inhibitor targets ubiquinone biosynthesis
This story highlights our collaborative study on the discovery and application of a new chemical probe to selectively inhibit human COQ8A in cells.
Cooperation
-
 Pantsar dynamics 01.10.2022 -
Pantsar dynamics 01.10.2022 - -
 Molecular Modeling and Drug Design Research Group 01.01.2010 -
Molecular Modeling and Drug Design Research Group 01.01.2010 - -
 Data-Driven Drug Design 01.01.2025 -
Data-Driven Drug Design 01.01.2025 - -
 Cancer Cell Plasticity - Ketola Lab 01.09.2019 -
Cancer Cell Plasticity - Ketola Lab 01.09.2019 -
Keywords
Leaders
Senior Researchers
Post-doctoral Researchers
-
Luca Pozzetti
Postdoctoral ResearcherSchool of Pharmacy, Faculty of Health Sciences -

Arun Tonduru
Postdoctoral ResearcherSchool of Pharmacy, Faculty of Health Sciences
Doctoral Researchers
-
Renne Leini
Doctoral ResearcherSchool of Pharmacy, Faculty of Health Sciences -
Tuomas Tikkanen
Doctoral ResearcherSchool of Pharmacy, Faculty of Health Sciences
Technicians
Other group members
-
Ella Wilska Project Researcher
Publications
39 items-
4-Anilinoquinazoline carbamate derivatization as a platform for prodrug design and localized drug delivery
Carpio, Mabel Barreiro; Walker, Coleman D; Fikes, Audrey; Laitinen, Tuomo; Valdes-Pena, M Alejandro; Gani, Osman; Mallory, Micah; Asquith, Christopher RM; Brudno, Yevgeny; Pierce, Joshua G, 2026, Bioorganic chemistry, 178, 109912. A1 Journal article (refereed), original research -
PIP4K2C: an emerging fulcrum for multiple diseases
Pozzetti, Luca; Mishra, Manjari; Einav, Shirit; Asquith, Christopher R. M., 2026, Nature reviews drug discovery, 25, 3, 166-166. B1 Non-refereed journal articles -
Medicinal chemistry applications of the Dimroth Rearrangement to the synthesis of biologically active compounds
Pozzetti, Luca; Pinhammer, Maja M; Asquith, Christopher RM, 2025, European journal of medicinal chemistry, 289, 117399. A2 Review article, Literature review, Systematic review -
Myeloid-specific CAMKK2 deficiency protects against diet-induced obesity and insulin resistance by rewiring metabolic gene expression and enhancing energy expenditure
Ortiz, Andrea R; Nay, Kevin; Stork, Brittany A; Dean, Adam M; Hartig, Sean M; Coarfa, Cristian; Tegegne, Surafel; Asquith, Christopher RM; Frigo, Daniel E; York, Brian; Means, Anthony R; Febbraio, Mark A; Scott, John W, 2025, Molecular metabolism, 101, 102250. A1 Journal article (refereed), original research -
PIP4K2C inhibition reverses autophagic flux impairment induced by SARS-CoV-2
Karim, Marwah; Mishra, Manjari; Lo, Chieh Wen; Saul, Sirle; Cagirici, Halise Busra; Gourdelier, Manon; Ghita, Luca; Ojha, Amrita; Tran, Do Hoang Nhu; Agrawal, Aditi; McGraw, Connor; East, Michael P; Gammeltoft, Karen Anbro; Sahoo, Malaya Kumar; Mooney, Nancie A; Johnson, Gary L; Das, Soumita; Leyssen, Pieter; Neyts, Johan; Chiu, Winston; Cohen, Courtney A; Bukh, Jens; Gottwein, Judith; Dye, John M; Neff, Norma; Jackson, Peter K; Pinsky, Benjamin A; Laitinen, Tuomo; Pantsar, Tatu; Poso, Antti; Za, 2025, Nature communications, 16, 1, 6397. A1 Journal article (refereed), original research -
Discovery and optimization of narrow spectrum inhibitors of Tousled like kinase 2 (TLK2) using quantitative structure activity relationships
Asquith, Christopher R M; East, Michael P; Laitinen, Tuomo; Alamillo-Ferrer, Carla; Hartikainen, Erkka; Wells, Carrow I; Axtman, Alison D; Drewry, David H; Tizzard, Graham J; Poso, Antti; Willson, Timothy M; Johnson, Gary L, 2024, European journal of medicinal chemistry, 271, 116357. A1 Journal article (refereed), original research -
Enhancing colistin efficacy against Salmonella infections with a quinazoline-based dual therapeutic strategy
Lobertti, Carlos A; Gizzi, Fernán O; Magni, Christian; Rial, Analía; Chabalgoity, José A; Yim, Lucía; Blancato, Víctor S; Asquith, Christopher R M; García Véscovi, Eleonora, 2024, Scientific reports, 14, 1, 5148. A1 Journal article (refereed), original research -
Pentathiepins are an understudied molecular prism of biological activities
Pozzetti, Luca; Asquith, Christopher RM, 2024, Archiv der pharmazie, 357, 12, 2400646. A2 Review article, Literature review, Systematic review -
TLK2: a target for cancer and viral latency
Pozzetti, Luca; East, Michael P; Laitinen, Tuomo; Asquith, Christopher RM, 2024, Nature reviews drug discovery, 23, 12, 886-886. B1 Non-refereed journal articles -
The Synthesis and Biological Applications of the 1,2,3-Dithiazole Scaffold
Kalogirou, Andreas S; Oh, Hans J; Asquith, Christopher RM, 2023, Molecules, 28, 7, 3193. A2 Review article, Literature review, Systematic review


